EXECUTIVE SUMMARY
Facebook sparked some buzz at the recent Digital Pharma East conference by sharing a case study on the first scrolling important safety information (ISI) in a Facebook post. Bayer BETASERON® and Facebook leveraged an existing format to create a video ad that featured the product name and indication in the text copy, with full ISI in the video.
As brands develop their 2017 plans, this strategy provides an additional option, as some brands may now be able to run full product promotions where they previously were only able to run reminder or reminder-like ads.
This POV provides background on the requirements for pharmaceutical advertising within social media and provides a breakdown of detailed information on the new use of scrolling ISI within Facebook posts, both paid and organic.
BACKGROUND
All pharmaceutical ads, including those in social media, must fall into one of a few categories:
- Disease awareness ads raise awareness about a condition but don’t mention or imply any specific drug.
- Reminder ads give the drug’s name but not its use.
- Reminder-like ads for black-box drugs include a link to the PI but are otherwise the same as reminder ads.
- Full product promotions mention the product name and condition and must include all of the following elements as described in the character-constrained draft guidance (which takes up a lot of space in the copy and is not always practical)
- Brand name
- Generic name and/or active ingredients
- Non-misleading indication statement
- All contraindications (other than mere hypersensitivity to the active ingredients) and life-threatening risks
- An abbreviated risk statement
- A link to full risk information (and additional elements required of a product promotion, such as dosage form and quantitative ingredient information)
The use of the scrolling ISI within Facebook’s video format could be used to accommodate all of the requirements listed above, providing an option for marketers to communicate information about a brand within the Facebook platform. Next, let’s take a closer look at how it was done.
FACEBOOK SCROLLING ISI: A CLOSER LOOK
At Digital Pharma East, Bayer’s Ozgun Demir, associate director of e-marketing, co-presented with Craig Hashi of the Facebook Health team to showcase a new Facebook advertisement for the BETACONNECT electronic autoinjector for multiple sclerosis. The team used an existing Facebook ad format that included scrolling ISI as a video that auto-plays while users scroll through their feeds. With the scrolling ISI constantly present in the ad, they were able to supply the FDA-required risk statement in a full product promotion.
As reported in FiercePharma, Hashi said, “While some pharmas already do that in banner ads, it had not been done inside Facebook ads until this campaign.”
Videos in Facebook posts can actually be up to 45 minutes in length. While that length is not recommended, it does allow plenty of time for a full ISI to be shown. And because these videos play in both desktop and mobile viewing, the ISI is served up across multiple types of devices.
See the scrolling ISI in action in this video captured by a conference attendee. Below, we provide more detail on the Bayer example.
THE BAYER BETASERON SCROLLING ISI FACEBOOK AD

This strategy can actually be applied in a variety of existing Facebook formats, including both organic and paid posts. Below, we compare and break down the two types.
Video ads do not live on the Facebook page timeline and only show in users’ News Feeds on desktop and mobile. Character limits are too strict for ISI.
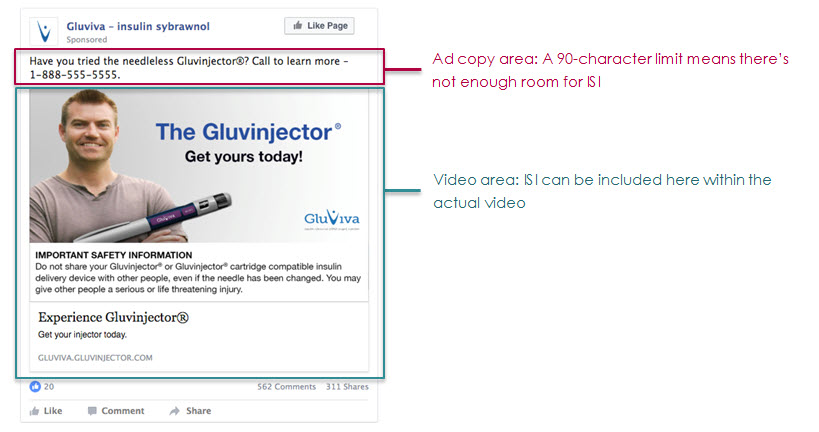
Organic posts live on the Facebook page timeline, as well as users’ News Feeds on desktop and mobile. These provide two options for featuring ISI, either in video or in copy, due to this format’s more generous character limit. Promoted posts work the same way.

REMAIN VIGILANT, STAY COMPLIANT
It’s worth noting that this approach doesn’t provide a blank check for compliance. Compliance will still largely depend on the overall messaging of each ad and the use of all of the post elements and how they work together. The Bayer ad, in fact, represented a lower-risk approach since the copy is actually promoting the BETACONNECT™ electronic autoinjector and avoids making strong product claims. We recommend clients refer to their own existing branded banner ad formats as a precedent for the approach to these types of Facebook ads.
It’s also important to understand that users can still comment or share posts on pages that have those functionalities enabled. So while any page can use this technique, Intouch only recommends it for whitelisted branded treatment pages (which have comments turned off)
and branded treatment pages that allow comments (and have procedures in place to monitor and moderate the page).
Of course, disease awareness pages avoid naming products by nature, so scrolling ISI would not be appropriate on posts from those pages. To create compliant links from disease awareness pages to branded destinations, marketers can still use workarounds through the use of tools
like ssshare.it.
CONCLUSION
Rumors from Facebook, the formation of a new Facebook Health team, and actions such as presenting at Digital Pharma East suggest Facebook might be interested in obtaining more pharma ad dollars and may offer more pharma-friendly ad formats in the future.
Note the ISI technique that Bayer/Facebook implemented involved simply taking an existing video ad format and placing scrolling ISI within the video. Essentially, this technique could be explored for use on additional video-enabled platforms such as Instagram (which is owned by Facebook) and others.
In the end, the fact that Facebook recognizes that pharma marketers face unique challenges and is helping the industry overcome those challenges is a sign they are willing to work with healthcare marketers. While this scrolling ISI ad format is not needed for all types of promotion, we always welcome more options that allow marketers to reach consumers with messages that are right for them.
To learn how Intouch can assist you with your pharma social media communities and advertising, please reach out to your Intouch representative.

